Zinc reacts with concentrated Nitric acid to produce Zinc nitrate, nitrogen dioxide and water..
Accordingly, why zinc does not react with nitric acid?
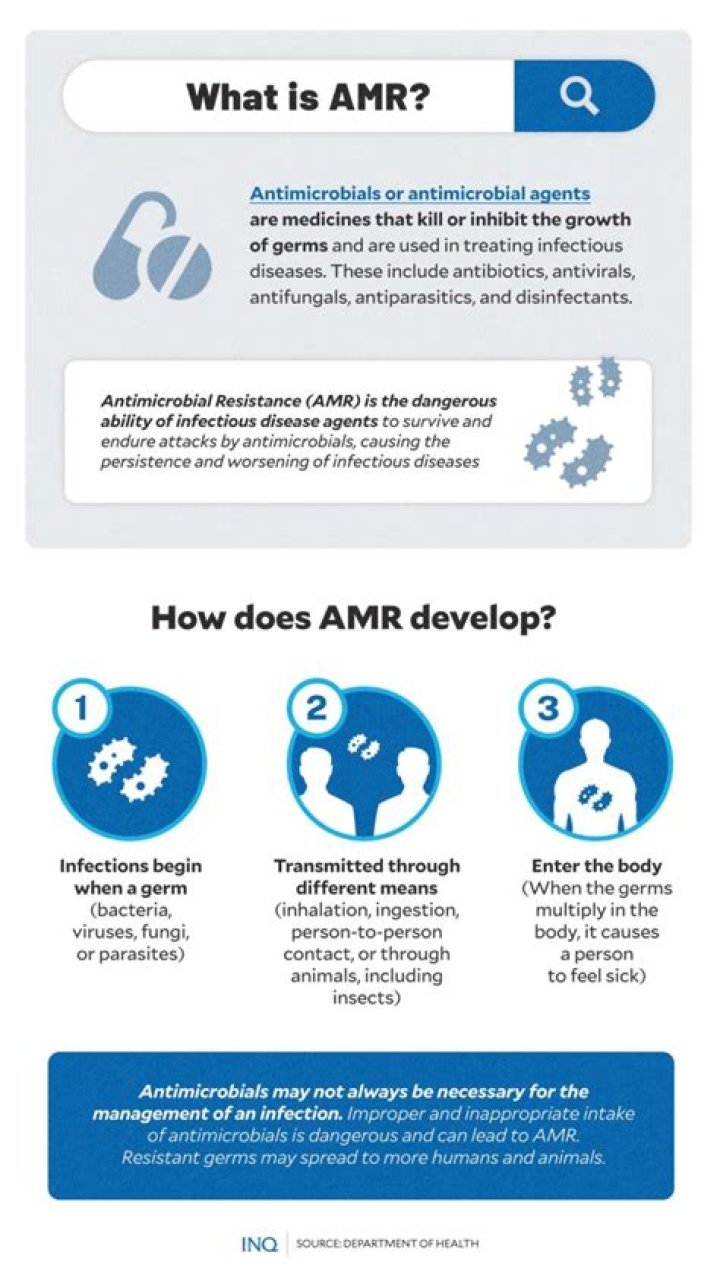
Aluminum and zinc do not react with concentrated nitric acid since a thick, hard to dissolve oxidation layer builds (passive oxidation) that protects the metal against further assaults.
Subsequently, question is, which acid does not react with zinc? If you use large zinc granules and cold dilute nitric acid, you might not observe much reaction at first. This is because of the formation of ZnO coating that prevents further reaction.
In this manner, does Zinc react with dilute nitric acid?
The products formed when zinc reacts with dilute nitric acid are zinc nitrate and hydrogen gas; i.e. 5Zn + 12HNO3 ----> 5Zn(NO3)2 + N2 + 6H2O. These products are formed due to the reduction of HNO3 by nascent hydrogen, which is produced in increasing amounts as the concentration of nitric acid is lowered; e.g.
What does zinc oxide and nitric acid make?
Zinc reacts with dilute Nitric acid to produce Zinc nitrate, nitric oxide, and water. 3Zn + 8HNO3 → 3Zn(NO3)2 + 2NO + 4H2O. Zinc reacts with concentrated Nitric acid to produce Zinc nitrate, nitrogen dioxide, and water.
Related Question Answers
What happens when zinc react with nitric acid?
Zinc reacts with concentrated Nitric acid to produce Zinc nitrate, nitrogen dioxide and water.Does zinc dissolve in nitric acid?
Zinc nitrate is usually prepared by dissolving zinc in nitric acid, this reaction is concentration dependent, with a reaction in concentrated acid also forming ammonium nitrate: On heating, it undergoes thermal decomposition to form zinc oxide, nitrogen dioxide and oxygen.What metal does not react with nitric acid?
Some precious metals, such as pure gold and platinum-group metals do not react with nitric acid, though pure gold does react with aqua regia, a mixture of concentrated nitric acid and hydrochloric acid.Does nitric acid dissolve copper?
Nitric acid not only acts upon copper, but it acts upon fingers. The pain led to another unpremeditated experiment.Does nitric acid dissolve nickel?
Here are several ways to dissolve nickel metal. Hydrochloric acid + hydrogen peroxide: Nickel dissolves with moderate speed in this mixture. Nitric acid: Nickel also dissolves well with this acid mixture. Nitrogen dioxide is produced as well as deliquescent nickel nitrate.What metals will not dissolve in nitric acid?
Noble Metals Gold, for example, does not even react with nitric acid, a strong oxidizing agent, although it will dissolve in aqua regia, a solution of concentrated nitric and hydrochloric acid.What happens when iron reacts with dilute nitric acid?
“Reacting iron powder with dilute nitric acid will produce Iron III Nitrate, nitrous oxide & water (the nitric acid oxidising the iron to the Fe3+ cation). The reaction proceeds slowly. The name of the non-metal comes from the anion in the acid.Does copper react with nitric acid?
Copper is an unreactive metal and doesn't react in normal circumstances with dilute acids. However, it does react with nitric acid. Nitric acid is an oxidizing agent and the reaction is not the usual acid + metal reaction. The products are oxides of nitrogen instead of hydrogen.What happens when copper reacts with nitric acid?
Copper and Nitric Acid. Nitric acid reacts with copper according to the reaction: 4 HNO3(l) + Cu(s) ==> Cu(NO3)2(s and aq) + 2 NO2(g) + 2 H2O(l) The copper nitrate salt that forms is a deep blue color. The nitrogen dioxide is a maroon vapor.What happens when zinc granules are treated with dilute solution of nitric acid?
Similarly, Hydrogen gas and Zinc Nitrate is formed when the Zinc granules are treated with HNO3 . In the case of NaCl, Zinc being less reactive than Sodium, no reaction occurs. When zinc is made to react with NaOH, it forms Sodium zincate and hydrogen gas.What happens when granulated zinc reacts with dilute Sulphuric acid?
When zinc granules react with dilute sulphuric acid then the product will form is zinc sulphate and removal of hydrogen gas.Why addition of zinc to dilute nitric acid is a chemical change?
Addition of zinc nitrate to water is a physical change as after dilution , the composition of the compound do not gets altered whereas the addition of zinc nitrate in dilute nitric acid gives two different compounds with different compositions .Can lead react with nitric acid?
Lead react with nitric acid to produce lead(II) nitrate, nitric oxide and water.What happens when zinc reacts with sodium hydroxide?
Zinc is a transition metal which shows moderate reactivity. Zinc reacts with NaOH to form sodium zincate with the evolution of hydrogen gas.Why hydrogen gas is not evolved when a metal reacts with nitric acid?
Hydrogen gas is not evolved when a metal reacts with nitric acid. It is because HNO3 is a strong oxidising agent. It oxidises the H2 produced to water and itself gets reduced to any of the nitrogen oxides (N2O, NO, NO2). But magnesium (Mg) and manganese (Mn) react with very dilute HNO3 to evolve H2 gas.Which gas is liberated when zinc reacts with dilute hno3?
hydrogen gas
When zinc reacts with dilute hno3 the oxidation state of nitrogen changes from?
Generally, when zinc reacts with nitric acid, it forms zinc nitrate. But when we take very dilute solution of nitric acid, if forms ammonium nitrate as well. So, here Nitrogen is reducing from +5 to +4.Does Zinc react with hydrochloric acid?
The metal zinc readily reacts with hydrochloric acid to produce hydrogen gas (H2) and zinc chloride (ZnCl2). The reagents are zinc and hydrochloric acid, and the sum is 0 + 2 * ( -167.2) = -334.3.What does zinc not react with?
Zinc is a fairly reactive metal that will combine with oxygen and other non-metals. Zinc metal tarnishes in moist air and burns in air to form the white zinc oxide. It is a fair electrical conductor and will react with dilute acids to release hydrogen. Zinc does not react with water.